Boosting butadiene yield at low temperature from ethanol upgrading over the ZnO/LaPO4 catalyst through HCO3⁻ modification
Abstract
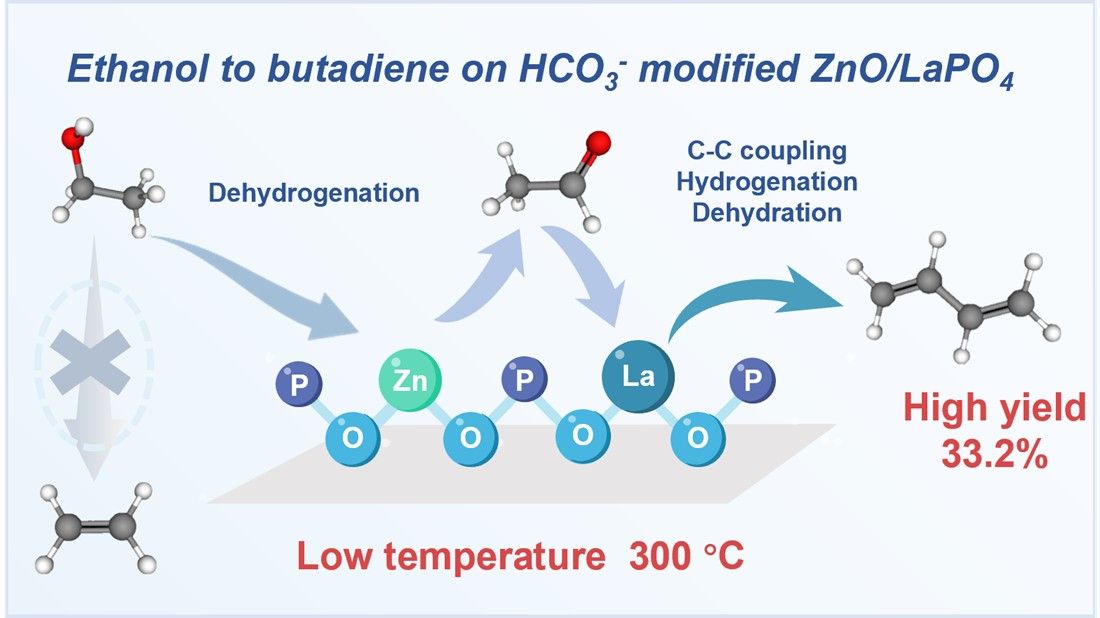
The catalytic conversion of ethanol to butadiene contributes to reducing dependency on petroleum sources, along with the advantages of low cost and sustainability. A significant challenge involves the competition between dehydrogenation and dehydration at high temperatures (≥ 350 °C), which suppresses the C-C coupling process. To address this issue, we design a HCO3⁻ modified lanthanum phosphate support that shows an improved surface area and reduced surface acidity. After loading zinc oxide, the obtained ZnO/LaPO4 catalyst efficiently converts ethanol to butadiene showing a yield of 33.2% with a weight hourly space velocity of 0.73·h-1 at 300 °C. The improved surface area of catalysts is beneficial for ethanol adsorption and diffusion, while the reduced surface acidity suppresses the ethanol dehydration reaction. As the highly dispersed dehydrogenation sites, [Zn-O-P] formed via phosphate coordination anchoring zinc oxide, resulting in efficient low-temperature ethanol activation. Kinetics experiments and in-situ diffuse reflectance infrared Fourier transform spectroscopy indicate that the pathway follows ethanol dehydrogenation to acetaldehyde over the [Zn-O-P] sites, while the surface exposed La3+ sites facilitate acetaldehyde coupling and dehydration to yield butadiene. The synergy between [Zn-O-P] and La3+ steers the reaction pathway towards carbon-chain growth, in turn suppressing the competitive direct dehydration of ethanol to ethylene/ether. Furthermore, the catalyst exhibits excellent reusability. This study provides a strategy for converting ethanol to butadiene at low temperatures.

