Thermally-driven non-catalytic reaction facilitating the activation of boron nitride for oxidative dehydrogenation of propane
Abstract
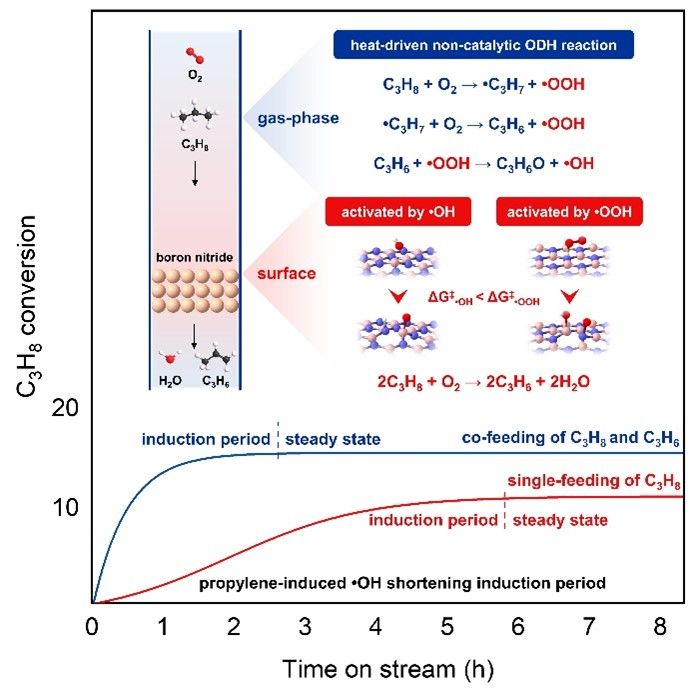
Boron nitride (BN) is active and selective catalyst for oxidative dehydrogenation (ODH) of propane to propylene. The in-situ generated boron oxyhydroxides during the induction period of reactions are recognized as the key active phase. However, the mechanism of them formation remains elusive because of the complexity of such ODH reaction involving surface and gas-phase free radical reactions. Herein, we reveal external thermally-driven non-catalytic ODH reaction as the initiation mechanism for BN activation during the induction period—a paradigm distinct from surface-mediated processes, by designing experiments with confined gas-phase space. Density functional theory (DFT) calculations reveal that the trace amount of H2O, hydroxyl radical (•OH), and hydroperoxyl radical (•OOH) formed during reaction can cleave the B-N bond of BN to generate boron oxyhydroxides, with a reactivity order of •OH> H2O> •OOH. Moreover, propylene can react with •OOH to form •OH, accelerating the oxidation process. Such viewpoint is supported by an observed short induction period in co-feeding propylene and propane experiments, and rapid boron oxyhydroxides formation in the ex/in-situ characterizations. In contrast, the formation of boron oxyhydroxides is inhibited in absence of the gas-phase space above the catalyst bed, regardless of propylene addition, confirming the essentiality of thermally induced gas-phase ODH reaction. Finally, a tandem system utilizing in-situ formed propylene was designed to shorten the induction period and promote propane conversion. This study unveils the active phase formation aspect on BN and offers alternative for designing efficient catalytic systems.

