C-S bonds modulated nanointerface tension to create stable magnetic hollow nanocarbons for efficient microplastics capture
Abstract
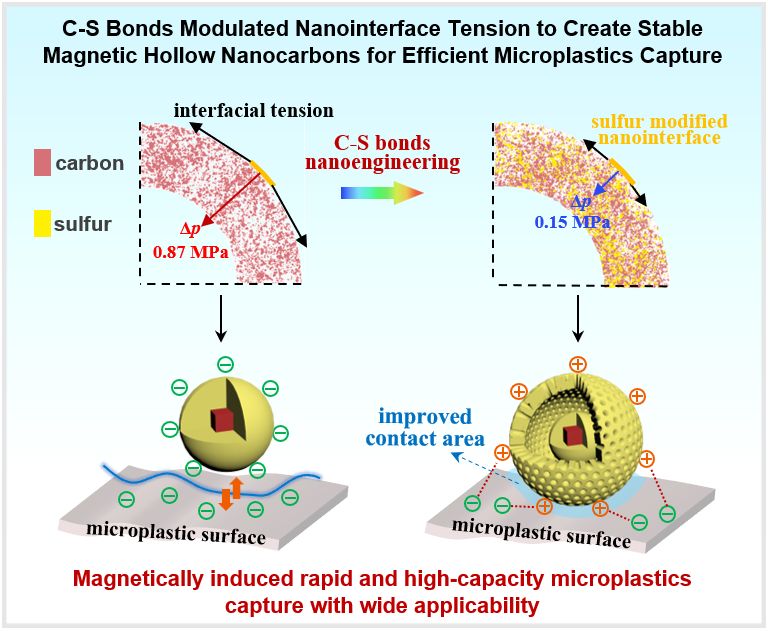
Microplastic pollution poses significant threats to aquatic ecosystems and human health. Hollow nanomaterials are promising adsorbents for microplastics remediation due to their tailorable architectures, functions, and large contact area. Nevertheless, the structural stability of well-defined nanostructures has always been a critical factor, and understanding the stability principle is desired. Herein, we fabricated magnetic hollow nanocarbons as “nano-analytical tool”, revealing that the stability is related to additional pressure caused by nanointerface tension at curved carbon shell surface. To mitigate this, we introduced C-S bonds by sulfurizing carbon matrix, suppressing the condensation of oxygen-containing groups and thereby reducing interface tension. As a showcase, the stable hollow Fe3O4@C/S enabled rapid and efficient microplastics capture (100% within 10 seconds, 53600 mg/g capacity) under an alternating magnetic field, owing to the magnetically accelerated mass transfer and increased contact area. Additionally, sulfur modification broadens applicability range where carbon surface is oppositely charged to microplastics, expanding the universality in capturing multiple types of microplastics, even under challenging conditions including different pH and salinities. This work offers guidance into the precisely synthesis of hollow nanomaterials from nanointerface perspective. The design principles involving sulfur modification and high-contact area may open prospects for high-capacity microplastics capture in complex aquatic environments.

